Natural Gas is Essential
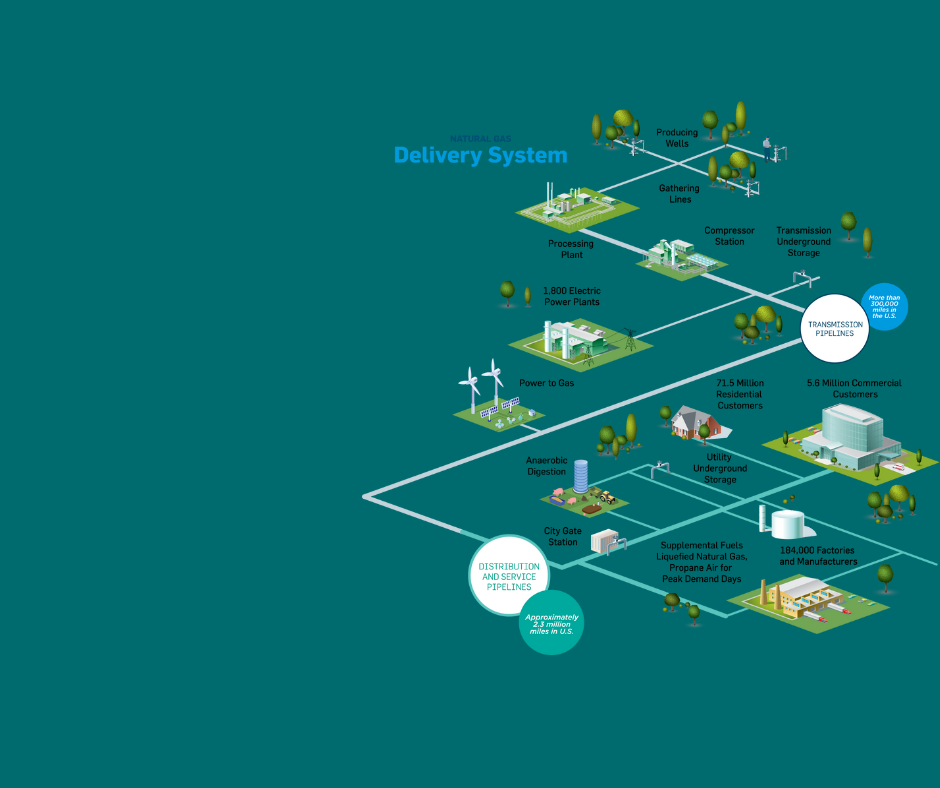
Natural gas is an essential part of America’s energy mix. Natural gas supplies nearly one-quarter of all the energy in the United States. AGA members distribute natural gas to homes, businesses, power plants, and factories.

Cleaner Energy
By the Numbers
189 Million
More than 189 million Americans and 5.8 million businesses use natural gas because it is affordable, reliable, safe and essential to improving our environment.
70%
Emissions from natural gas distribution systems have dropped 70% since 1990.
1+ per minute
More than one new residential customer signs up for natural gas service every minute on average, and approximately 60 businesses begin new natural gas service every day.
AGA Playbook
Featured Resource
The AGA Playbook tells the story of an industry delivering the reliable energy that Americans depend upon. AGA invites customers, policymakers, and anyone invested in our clean energy future to flip through the pages of the AGA Playbook to learn more about the commitment of America’s natural gas utilities to affordability, the environment, innovation and safety.

Environment
AGA’s Climate Change Commitment
AGA is committed to reducing greenhouse gas emissions through smart innovation, new and modernized infrastructure and advanced technologies. This pledge includes 10 commitments to further reduce methane emissions from natural gas utility systems. It also includes eight principles for an effective national policy approach to reducing greenhouse gas emissions and addressing climate change.